 -(Rubidium Atomic Structure, 2015)
-(Rubidium Atomic Structure, 2015)
Rubidium was discovered by Robert Bunsen and Gustav Kirchhoff as an impurity associated with samples of the mineral lepidolite in Germany, 1861. It is found in some potassium minerals (lepidolites, biotites, feldspar, carnallite), sometimes with caesium as well. The origin of the name rubidium comes “from the Latin word rubidius meaning dark red or deepest red.”-(Rubidium: The Essentials, 2015). The name was coined due to its (rubidium’s) bright red spectroscopic lines. “Rubidium has no biological role but is said to stimulate the metabolism.”-(Rubidium: The Essentials, 2015). Because Rubidium is one of the most electropositive and alkaline elements, it ignites spontaneously in air and reacts violently with water- meaning it cannot be found as a free metal in nature. It alloys with gold, caesium, sodium, and potassium. Although Rubidium is rare, it is the 16th most abundant element in the earth's crust. –(Rubidium: The Essentials, 2015).
Rubidium is mostly obtained as a by-product of refining lithium. “Rubidium is used in vacuum tubes as a … material that combines with and removes trace gases from vacuum tubes.” –(It's Elemental - The Element Rubidium, 2015).
Difficulty with pure rubidium production is Rubidium and Cesium, as well as other alkali metals, are always found together in nature and it is difficult to handle because “it ignites spontaneously in air, and it reacts violently with water.” –(rubidium (Rb) | chemical element | Encyclopedia Britannica, 2015).
Rubidium is very reactive because it has only one electron in it’s outer shell. This makes it want to bond with other elements and lose that electron. An increased desire to bond means more reactivity. -(Chem4Kids.com: Elements & Periodic Table: Alkali Metals, 2015).
Future uses for Rubidium could be manufacturing photocells and it could be used as a propellant in ion engines on spacecraft. –(It's Elemental - The Element Rubidium, 2015).
QUICK FACTS
o Symbol: Rb
o Atmoic number: 37
o Relative atomic mass: 85.4678
o Standard state: solid at 24.85°C
o Colour: silvery white
o Classification: Metallic
o Group in periodic table: 1, Alkali metal
o Period in periodic table: 5
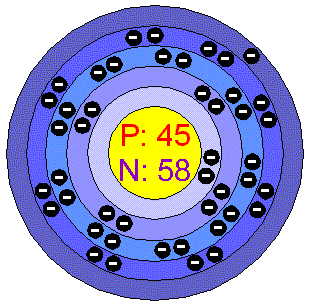
o Electron shell structure: 2.8.18.8.1
o Density of solid: 1532 kg m-3
o Melting point: 39.31 °C
o Boiling point: 688 °C
-(Rubidium: The Essentials, 2015).
Rubidium is mostly obtained as a by-product of refining lithium. “Rubidium is used in vacuum tubes as a … material that combines with and removes trace gases from vacuum tubes.” –(It's Elemental - The Element Rubidium, 2015).
Difficulty with pure rubidium production is Rubidium and Cesium, as well as other alkali metals, are always found together in nature and it is difficult to handle because “it ignites spontaneously in air, and it reacts violently with water.” –(rubidium (Rb) | chemical element | Encyclopedia Britannica, 2015).
Rubidium is very reactive because it has only one electron in it’s outer shell. This makes it want to bond with other elements and lose that electron. An increased desire to bond means more reactivity. -(Chem4Kids.com: Elements & Periodic Table: Alkali Metals, 2015).
Future uses for Rubidium could be manufacturing photocells and it could be used as a propellant in ion engines on spacecraft. –(It's Elemental - The Element Rubidium, 2015).
QUICK FACTS
o Symbol: Rb
o Atmoic number: 37
o Relative atomic mass: 85.4678
o Standard state: solid at 24.85°C
o Colour: silvery white
o Classification: Metallic
o Group in periodic table: 1, Alkali metal
o Period in periodic table: 5
o Electron shell structure: 2.8.18.8.1
o Density of solid: 1532 kg m-3
o Melting point: 39.31 °C
o Boiling point: 688 °C
-(Rubidium: The Essentials, 2015).
